
In short, all of these forms the geometry of the Sulfur Fluorine molecule. Keeping this in consideration, how is sf2 polar? 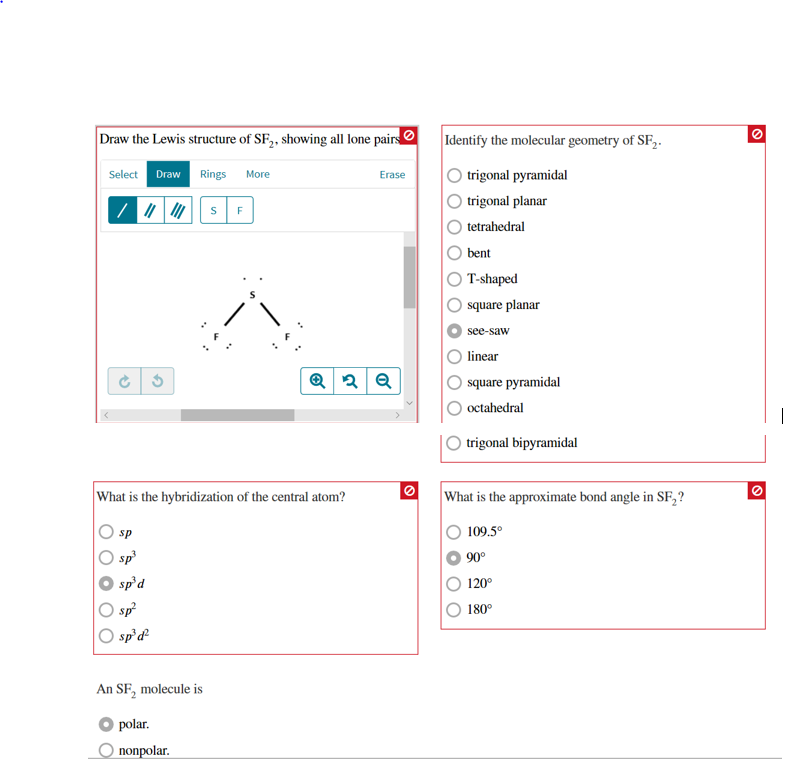 
Because the SF2 molecule is not symmetrical there is a region of unequal sharing. Polarity results from an unequal sharing of valence electrons. To determine if SF2 is polar we need to look at the molecular geometry or shape of the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
